Журнал "Макрогетероциклы"
Navigation
News
Impact Factor 2021 = 1.200 has been issued by ISI Web of Knowledge (JCR 2021).
Search
ISSN 1998-9539
Proton Exchange Involving Water Molecules and Internal NH Groups of Porphyrins and Chlorins in C6D6 Medium Based on DOSY Data
Dmitriy V. Belykh,a@ and Alexander L. Stolypkob
aInstitute of Сhemistry of Komi Scientific Centre of the Ural Branch of the Russian Academy of Sciences, 167000 Syktyvkar, Russia
bSyktyvkar State University, 167000 Syktyvkar, Russia
@Corresponding author E-mail: belykh-dv@mail.ru
DOI: 10.6060/mhc190444b
Macroheterocycles 2019 12(4) 392-397
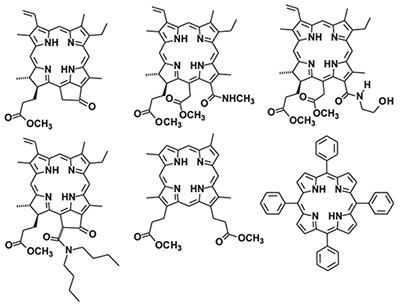
The processes of proton exchange involving water molecules and internal NH protons of a number of porphyrins and chlorins in a C6D6 medium were studied by DOSY method. It has been shown that the processes of proton exchange between the internal NH-groups of porphyrins and water molecules in the C6D6 medium occur similarly to the exchange in the CDCl3 medium. The differences observed can be explained by a significant increase of porphyrin-water associates lifetime in the C6D6 medium compared to CDCl3. A number of features of the participation of internal NH-groups of porphyrins with various structures in exchange processes were found. It has been shown that in a meso-substituted porphyrin macrocycle the exchange processes involving intracyclic NH groups are less intense than in the case of other porphyrins and chlorins. Based on a comparison of the kNH values (rate constant for the transfer of intracyclic NH protons of porphyrin to water molecules), for all chlorophyll derivatives, regardless of the presence of an exocycle in the molecule, the NH proton of the pyrrole ring III is much more actively involved into exchange processes than the NH proton of the pyrrole ring I. This, apparently, is due to the fact that in both cases the exchange of NH protons with free water occurs through a water molecule associated with porphyrin. At the same time, the lifetime of the porphyrin-water associate for pyrrole ring III significantly exceeds the same time for pyrrole ring I, which leads to an increase in the probability of exchange for pyrrole ring III.
| Attachment | Size |
|---|---|
| mhc190444b.pdf | 625.05 KB |
- 1061 reads
- Русский
