Журнал "Макрогетероциклы"
Навигация
Новости
Web of Knowledge определил Импакт фактор журнала Макрогетероциклы за 2021 год: 1.200 (JCR 2021).
Поиск
ISSN 1998-9539
Релаксация возбужденных состояний в ковалентно-связанных диадах и триадах на основе триптофана, дейтеропорфирина и хинона
Э. И. Зенькевич,a@ Е. А. Ларкина,b Н. В. Коновалова,b А. П. Ступакc
aБелорусский национальный технический университет, 220013 Минск, Беларусь
bМИРЭА – Российский технологический университет, Институт тонких химических технологий им. М.В. Ломоносова, 119571 Москва, Россия
cИнститут физики им. Б.И. Степанова НАН Б, 220072 Минск, Беларусь
@E-mail: zenkev@tut.by
DOI: 10.6060/mhc181224z
Макрогетероциклы 2019 12(1) 47-57
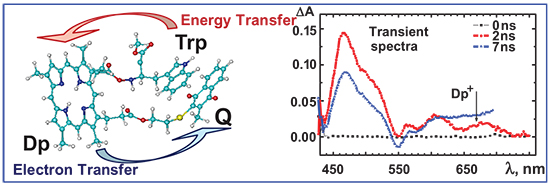
Для диад и триад, состоящих из дейтеропорфирина IX и хинона или одной либо двух молекул триптофана, ковалентно присоединенных по остаткам пропионовой кислоты в b-положениях порфирина, выполнены измерения стационарных и время-разрешенных спектрально-флуоресцентных характеристик в растворах различной полярности при 295 К. Для диад триптофан-порфирин было показано, что экспериментальные (FПЭэкспер = 0.75) и теоретические (FПЭтеор = 0.87) значения эффективности переноса энергии находятся в разумном соответствии, и теория индуктивного резонанса Ферстера применима для слабо взаимодействующих донорно-акцепторных систем на расстояниях RДА » 19¸25 Å. Для диады порфирин-хинон и триады триптофан-порфирин-хинон в диметилформамиде при 293 К обосновано, что тушение флуоресценции порфирина может быть адекватно описано полуклассической теорией Маркуса как эндотермический или слабо экзотермический неадиабатический фотоиндуцированный перенос электрона, происходящий в «нормальной» области с вероятностью kФПЭ = 2.7×108 с-1. На основании количественного экспериментального и теоретического анализа процессов переноса энергии и фотоиндуцированного переноса электрона обосновано, что формирование свернутых конформаций диад и триад маловероятно в жидких растворителях при комнатной температуре.
С полным текстом статьи можно ознакомиться на английской версии сайта
- 1084 просмотра
- English
